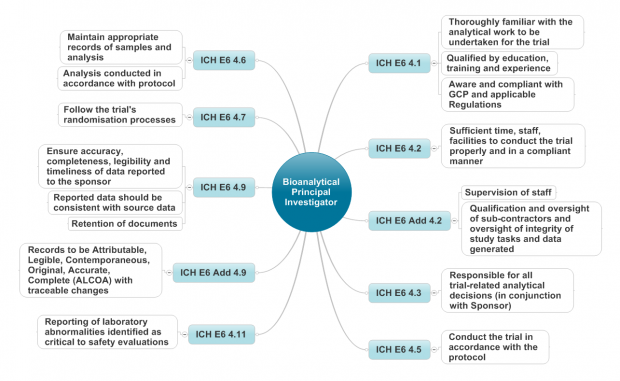
Updated data integrity requirements for GLP Monitoring Programme members

Members should conduct a documented review of their existing data integrity processes against the new OECD document to ensure that they fully comply and any required changes or additions are implemented.





Recent Comments